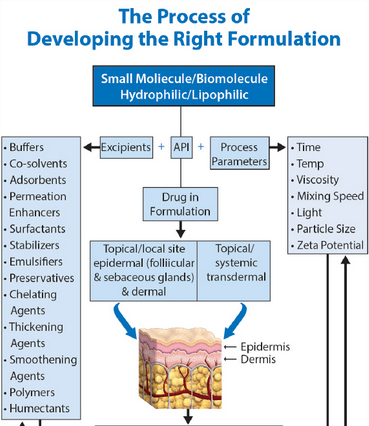
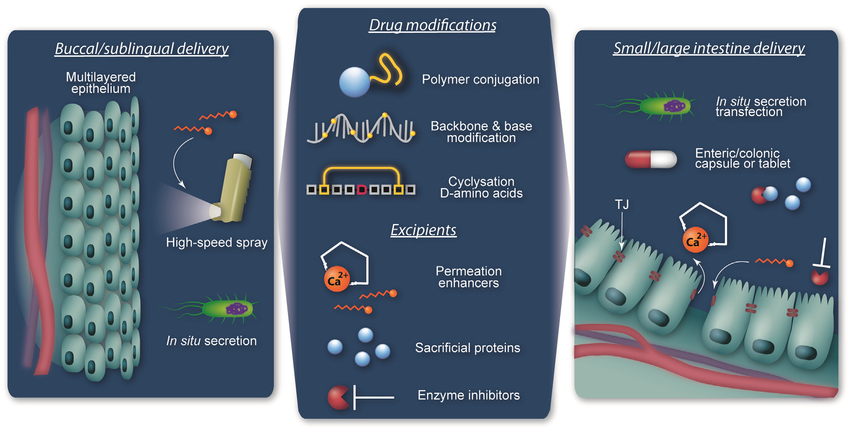
Brilacidin, computationally designed as a biomimetic polymer with a robust structure, and unlike peptide-based small molecules (an overview), is not subject to most of the traditional shortcomings of antimicrobial peptide (AMP)-based therapeutics, including toxicity and rapid proteolytic degradation. This can make drug formulation work, particularly the oral delivery (pdf) of peptidic medications, for example across the human intestine, more challenging (e.g., see the FORMAMP Project).
Brilacidin already has been shown to have a favorable safety profile and has met efficacy endpoints when administered systemically, via intravenous (IV) dosing, in a Phase 2b trial in Acute Bacterial Skin and Skin Structure Infection (ABSSSI), as well as when topically-applied -- as shown in Phase 2 trials in Ulcerative Proctitis / Ulcerative Proctosigmoiditis (UP/UPS) (administered in water as a retention enema) and in Oral Mucositis (OM) (given as a "swish and spit").
Future Brilacidin formulation development plans include foam and/or gel for the treatment of UP/UPS, and pills for the oral dosing of Ulcerative Colitis and Crohn’s Disease.
Physicians typically prefer topical therapies, over systemic ones, as they have safer profiles, are more conveniently administered ("patient-friendly"), and help ensure better compliance to treatments.
"While efficacy and safety often had greater relative importance than treatment process, treatment process also had a quantifiable impact on preference. In some instances, particularly in diabetes and autoimmune disorders, treatment process attributes had greater relative importance than some or all efficacy and safety attributes." (Source: "Preference for Pharmaceutical Formulation and Treatment Process Attributes." Patient Prefer Adherence. 2016; 10: 1385–1399.)
Source: "Oral Delivery of Macromolecular Drugs: Where We Are After Almost 100 Years of Attempts." Advanced Drug Delivery Reviews. Volume 101, 1 June 2016, Pages 108-121.