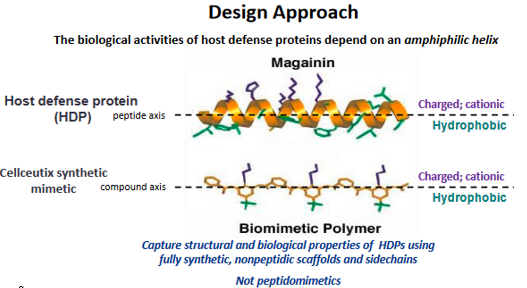
Brilacidin is a small, non-peptidic, fully synthetic mimic of Host Defense Proteins (HDPs), specifically defensins (pdf), that can be developed as systemic or topical agent.
An arylamide foldamer, and unlike most peptidic-based small molecules (e.g., Pexiganan), Brilacidin is not subject to the traditional shortcomings of antimicrobial peptide (AMP)-based compounds, including rapid proteolytic degradation. Instead, by using sophisticated coarse-grain computer modeling that mimicked the behavior of natural defensins (electrostatics, lipophicility, etc.), it was designed (pdfs) to be smaller (one-tenth the size) and then fine-tuned to exhibit enhanced pharmacological properties—more easily and much less expensively synthesized, more stable (a rigid backbone), more potent (by a 100-fold) and more selective (by a 1000-fold).
This biocomputational aspect of Brilacidin’s development (de novo design) has resulted in the drug candidate having much better exposure and efficacy in terms of its pharmacokinetics.
Beyond its antimicrobial properties—which itself can play a major role in treating certain inflammatory diseases given the association between pathogens and host organisms, inflammation and infection (e.g., see the link between Enterobacteriaceae and IBD; the link between Crohn’s and E. coli [against which HDP-Mimetics have shown activity]; and the link between Crohn’s and infections, including fungal ones—Brilacidin functions through the cyclic AMP/cyclic GMP pathways to suppress pro-inflammatory cytokines and chemokines, such as TNF-α, IL-1β, IL-6, IL-8, MIP2-α, MCP-1 and MMP-9.
Brilacidin’s inhibition of PDE4—a predominant phosphodiesterase expressed in neutrophils, T cells, macrophages and keratinocytes—leads to an increase in the intracellular cAMP concentration, thereby reducing the production of pro-inflammatory mediators and increasing anti-inflammatory mediators.
Additional research is underway to determine Brilacidin's potential effect on IL-17, a central driver of most inflammatory diseases, including many skin diseases.
Linked to below is a Nature Reviews Drug Discovery series on the IL-17 pathway.
Targeting the IL-17 Pathway in Inflammatory Diseases (poster, pdf)