A recent Bloomberg article paints a rosier picture for 2017 as to Biotech M&A activity and dealmaking: “Pharma is a furnace that constantly needs drug candidates to replace failed projects and expired patents, and baby biotechs are the coal.”
Sentiment conveyed elsewhere as well:
"Pharmaceutical Firms Braced for a Year of Rampant M&A." (The Telegraph)
"Mergers and Acquisitions Among Biopharma Companies Expected to Soar This Year." (Managed Care)
In 2016, 326 deals were completed, worth approximately $91 billion, down from $118 billion the year prior.
A surplus of cash and marketable securities, in excess of $220 billion according to Bloomberg, as well as talk of the Trump Administration incentivizing pharmaceutical companies to repatriate overseas money, should serve to spur dealmaking activity this year.
As conveyed by Innovation management in a presentation given at the 2017 Biotech Showcase held in San Francisco, CA, the company’s focus this year is on partnerships—identifying new opportunities as well as continuing discussions with companies already under Confidential Disclosure Agreement (CDA).
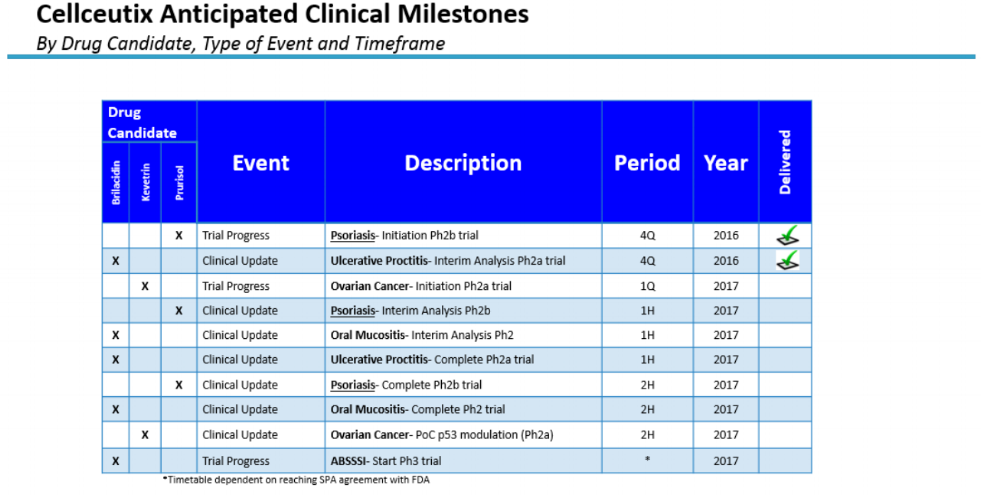
With a number of upcoming anticipated clinical milestones (see below), management is hopeful that partnerships can be struck in 2017 to help advance the Company's pipeline of First-in-Class drug candidates—Prurisol, Kevetrin and Brilacidin—toward market approval.
Source: https://www.bloomberg.com/gadfly/articles/2017-01-03/biotech-m-a-in-2017-a-new-hope